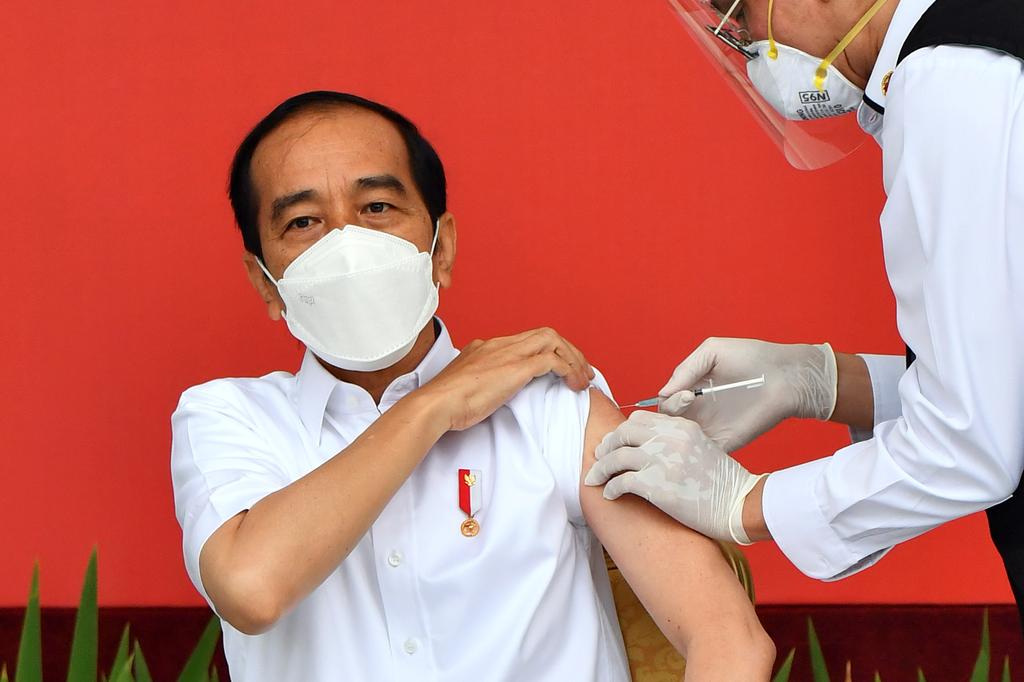
JAKARTA -- Indonesia launched one of the world’s biggest COVID-19 vaccination campaigns on Wednesday with President Joko Widodo getting the first shot of a Chinese vaccine as his country fights one of the worst coronavirus outbreaks in Asia.
The drive aims to inoculate 181.5 million people, with the first to be vaccinated receiving the CoronaVac vaccine from China’s Sinovac Biotech, which Indonesia authorised for emergency use on Monday.
Dressed in a white shirt and wearing a mask, the president, who is known as Jokowi, got his shot at the presidential palace.
“Vaccination is important to break the chain of COVID-19 transmission and give protection to us and safety to every Indonesian and help accelerate economic recovery,” Jokowi said after getting his injection.
Some other officials being vaccinated showed off their shot marks to waiting journalists and flexed their arms.
Minister of Health Budi Gunadi Sadikin has said nearly 1.5 million medical workers would be inoculated by February, followed by public servants and the general population within 15 months.
Unlike many countries, Indonesia intends to inoculate its working population first, rather than the elderly, partly because it does not have enough data from clinical trials on CoronaVac’s efficacy on older people.
Indonesia on Tuesday reported a daily record 302 coronavirus deaths, taking fatalities to 24,645. Its infections are at their peak, averaging more than 9,000 a day, with 846,765 total cases.
Indonesia’s stocks have risen in the last few days, helped by the launch of vaccinations, with the main index opening up around 0.7% on Wednesday.
“Vaccinations contributed a fairly positive market sentiment,” said Hans Kwee, director at investment manager Anugerah Mega Investama.
‘Not one bullet’
Southeast Asia’s largest economy suffered its first recession in more than two decades last year due to pandemic, with the government estimating a contraction of as much as 2.2%.
The government has said two-thirds of the 270 million population must be vaccinated to achieve herd immunity, with the cost of the programme expected to be more than 74 trillion rupiah ($5.26 billion).
Olivia Herlinda, a researcher at the Center for Indonesia’s Strategic Development Initiatives, said authorities had not taken into account the vaccine efficacy and virus reproduction rate to justify its herd immunity focus.
Epidemiologist Masdalina Pane said that vaccines had to be accompanied by increased testing and tracing.
“There’s not one bullet,” she said.
Budi said Indonesia’s testing and tracing needed improving, adding there was an imbalance in testing resources across the archipelago.
Indonesia has said its trials showed CoronaVac has an efficacy rate of 65.3%, but Brazilian researchers said on Tuesday the vaccine was only 50.4% effective.
Indonesia’s food and drugs authority BPOM did not immediately respond to a request for comment.
Bambang Heriyanto, corporate secretary of Bio Farma, the Indonesian company involved in the trials, said the Brazilian data was still above the 50% benchmark set by the World Health Organization.
Turkish researchers said in December CoronaVac showed a 91.25% efficacy based on analysis.
Indonesia expects to get another 122.5 million doses of CoronaVac by January 2022, with about 30 million doses due by the end of the first quarter this year.
It has also secured nearly 330 million doses of other vaccines, including from AstraZeneca and Pfizer and its partner BioNTech.
($1 = 14,060.0000 rupiah)