Vietnamese State President Nguyen Xuan Phuc has asked health authorities to promptly authorize Nano Covax, a vaccine being developed domestically, to make the first made-in-Vietnam vaccine available soon amid surging COVID-19 cases.
The president made the direction during a meeting at Ho Chi Minh City-based Nanogen Pharmaceutical Biotechnology JSC, which he visited on Thursday to review its production progress.
At the meeting, President Phuc emphasized the importance of the compliance with epidemic prevention and control rules and the acceleration of mass vaccination to prevent the rising coronavirus spread.
The leader said the Party and State have been proceeding with securing vaccine supplies from abroad while speeding up domestic vaccine research and production, with a vision to ensure proactiveness in inoculation.
Administrative procedures should be sped up so that the vaccine can be approved for emergency use soon provided it is safe for people, the president said.
The State leader added he set out a policy for domestic research on COVID-19 vaccine as early as March 2020 and directed the transfer of VND200 billion (US$8.7 million) to the health sector for vaccine studies.
As the pandemic has been raging in Vietnam, the production of a COVID-19 vaccine has become an urgent requirement that needs to be met soon, President Phuc said.
Ho Nhan, Nanogen director, told the president that after seven months of human trials since December 2020, the vaccine showed no adverse responses while having good immunogenicity and neutralization efficacy, including for the Delta variant of the coronavirus.
During the clinical trials on 14,000 people, the ratio of people having side effects was very low while no severe reactions or deaths were recorded, Nhan claimed.
About 30 countries have contacted the company for vaccine technology transfer negotiation, including India, the director told the meeting, adding that the World Health Organization (WHO) is also evaluating Nanogen's vaccine profile to integrate it into the world’s vaccine sources.
|
|
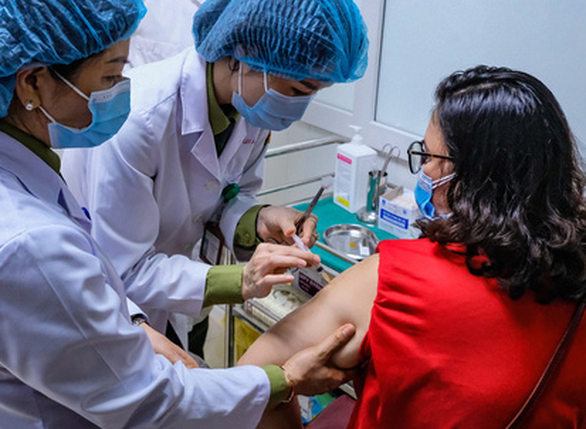
| This image shows a woman receiving a Nano Covax vaccine shot in phase-2 human trials conducted by Nanogen in Long An Province, Vietnam. Photo: Nam Tran / Tuoi Tre |
Nhan also announced that Nanogen is now able to produce 10 million Nano Covax doses per month and will increase the capacity to 30 million jabs in October.
The director proposed that Nagogen be granted an urgent conditional approval within this week or next week while the company is continuing phase-3 clinical trials.
President Phuc asked the health ministry and other relevant agencies to support the company in accelerating its vaccine production, and streamline administrative formalities in approval, on the principle of protecting vaccinated people.
“Based on the objective scientific assessments, the health ministry is requested to grant an early license to Nanogen so that the company can produce the vaccine and put it to use soon," he said.
"Safety is above all, but procedures should be quick in the current urgent context."
The Department of Science, Technology, and Training and the Pharmaceutical Department under the health ministry should push administrative procedures related to COVID-19 vaccine, while Nanogen should continue perfecting its vaccine product, the leader directed.
“We must take action faster, stronger, and more drastically as the pandemic is putting everyone in danger,” the president emphasized.
Vietnam needs to have 150 million vaccine doses to inoculate 70 percent of its 98 million-strong population to reach herd immunity, but the country has so far received only about 15 million shots in total from different sources, including COVAX Facility, contractual purchases, and donations, the health ministry said.
As of Friday, more than 5.5 million vaccine shots have been administered nationwide, with the numbers of people receiving the first and second doses nearing five million and some 546,000, respectively, the ministry reported.
Like us on Facebook or follow us on Twitter to get the latest news about Vietnam!